


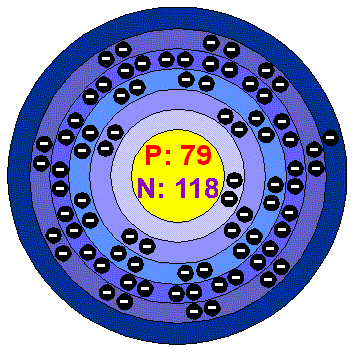
Number of Energy Levels: 6
Electron Configuration:
1s2.2s2.2p6.3s2.3p6.3d10.4s2.4p6.4d10.5s2.5p6.4f14.5d10.6s1
Shell Structure:
2.8.18.32.18.1
![]()
Gold was one of the first metals to be discovered by man in around 5000 BC, along with copper.
Gold can be found free as nuggets in quartz rock formations or as flakes and grains in sand.
Two thirds of the world's supply comes from South Africa, and 2/3 of USA production is from South Dakota and Nevada. Gold is found in sea water, but no effective economic process has been designed so far to extract it from this source.
It is yellow in color and is a soft, ductile, malleable metal that can be drawn into thin threads or hammered into very thin gold leaves that are 0.00002 mm thick. One ounce of gold can be beaten into a sheet covering over 9 square meters. One ounce of gold can also be drawn into 100 km of thin gold wire, which is used to make electrical contacts.
The most common use of pure gold is gold leafing.
Gold that is used in jewelry, coins, and dentistry is alloyed with other metals to give hardness. Pure gold content is rated in carats. 24 carats is pure gold.
It is estimated that all the gold in the world, so far refined, could be placed in a single cube 60 ft. on a side.
The chemical symbol for gold is Au, which is derived from the Latin word “aurum,” meaning 'shining dawn'. The Roman goddess of dawn was named Aurora, a name also derived from this term.
Gold is referred to as a noble metal, due to its excellent chemical stability.
Gold is an excellent conductor of heat and electricity, which is why is it used for semi-conductors in electronics and connectors in computer technology.
Gold is corroded only by a mixture of nitric and hydrochloric acid. In everyday use gold does not tarnish. Gold only dissolves in cyanide.
Hardness, which is defined as the ability of a material to resist surface abrasion, is assessed using a list of elements put in order such that each material is able to scratch any one below it. The highest hardness index is 10 and the lowest is 1. Gold is a soft metal, with a hardness index of 2.5 to 3.
Alchemy is an ancient branch of science in which people tried to turn other elements into gold.
Reactions of gold:
Under normal conditions, gold is stable in air. It dissolves in aqueous cyanide solutions in the presence of air.
Gold does not react with water
Gold metal reacts with chlorine, Cl2, or bromine, Br2, to form the trihalides gold(III) chloride, AuCl3, or gold(III) bromide, AuBr3, respectively.
On the other hand, gold metal reacts with iodine, I2, to form the monohalide gold(I) chloride, AuI.
2Au(s) + 3Cl2(g) 2AuCl3(s)
2Au(s) + 3Br2(g) 2AuBr3(s)
2Au(s) + I2(g) 2AuI(s)
Gold metal dissolves in aqua regia (the name of which means the "king of metals" was coined by alchemist because it was able to dissolve gold), a mixture of hydrochloric acid, HCl, and concentrated nitric acid, HNO3, in a 3:1 ratio.
Gold does not react with aqueous bases.