Name:
Copper
Symbol:
Cu
Atomic
Number:
29
Atomic
Mass:
63.546 amu
Melting
Point:
1083.0 °C (1356.15 °K, 1981.4 °F)
Boiling
Point:
2567.0 °C (2840.15 °K, 4652.6 °F)
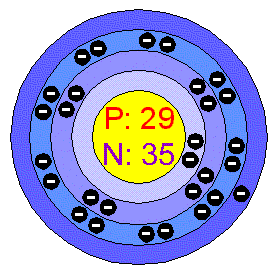
Number of
Protons/Electrons:
29
Number of
Neutrons:
35
Classification:
Transition Metal
Crystal
Structure:
Cubic
Density @ 293 K:
8.96 g/cm3
Color:
red/orange
Number of Energy Levels:
4
Number of Electrons per
Energy Level
First Energy Level:
2
Second Energy Level:
8
Third Energy Level:
18
Fourth Energy Level:
1
Electron Configuration:
1s2.2s2.2p6.3s2.3p6.4s1.3d10
Shell structure:
2.8.18.1

-
Copper is a fairly
abundant element. It occurs free in nature to some extent, but most copper is
extracted from ores -
in oxide ores as cuprite
and in sulphide ores as chalcocite, chalcopyrite, and bornite. It is found
chiefly in the Ural Mountains of Sweden and near Lake Superior in North
America.
-
It is a soft, ductile,
malleable metal, reddish brown in color.
-
Copper was first used by
man over 10,000 years ago. The oldest known copper finding is a copper pendant
that was discovered in what is now northern
Iraq and has been dated about 8700
B.C.
-
By 3000 B.C., copper was
beginning to be alloyed with arsenic and tin. For many centuries, bronze, an
alloy of copper and tin, was used almost exclusively for plows, tools,
weapons, armor, and decorative objects. The
Bronze Age, as the period of time
was known, ended in around 1200 BC.
-
With the invention and
commercialization of the telephone system in the 1870’s, copper grew in demand
as
the growing electric power grid began to consume large
quantities of copper wire.
-
The name copper is
derived from the Latin word “cyprium,” after the Island of Cyprus, located off
the coast of Greece.
-
Copper ore is mined from
open pits or underground mines. The Mine ore is crushed, ground, and
concentrated. The copper is run through furnaces several times, until the
non-copper elements are burned off or separated, resulting in “blister,” which
is a 99 % pure molten copper. Blister is poured into anode mold, containing an
electrolyte of sulfuric acid, copper sulfate and plating reagents. The result
is 99.9999 percent pure copper.

Statue of Liberty
- The Statue of Liberty
relies on copper! The weathering and oxidation of the copper skin of the
Statue has amounted to just .005 of an inch in a century. When she was
renovated for her centennial, the only copper part of the Statue that needed
refurbished was the torch, which was rebuilt with new copper and repainted to
match the green of the existing copper.
This
beautiful color results from contact with the moist atmosphere. Copper was
also used in the inner restoration, forming the structural skeleton and the
rivets that hold it all together.