

Name: Cadmium
Symbol: Cd
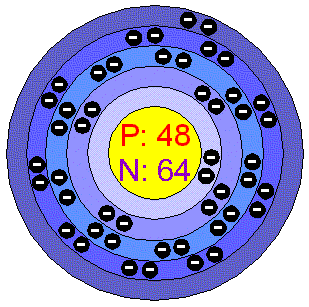
Atomic Number: 48
Atomic Mass: 112.411
amu
Melting Point: 320.9 °C
(594.05 °K, 609.62 °F)
Boiling Point: 765.0 °C
(1038.15 °K, 1409.0 °F)
Number of Protons/Electrons:
48
Number of Neutrons: 64
Classification:
Transition Metal
Crystal Structure:
Hexagonal
Density @ 293 K: 8.65
g/cm3
Color: Silvery
-
 Cadmium was
discovered by Stromeyer in 1817 as an impurity in a zinc carbonate ore. Stromeyer noted the yellow color of a specific batch of cadmia and found
cadmium as the contaminant.
Cadmium was
discovered by Stromeyer in 1817 as an impurity in a zinc carbonate ore. Stromeyer noted the yellow color of a specific batch of cadmia and found
cadmium as the contaminant.
- Cadmium itself is a
soft bluish-white metal which is easily cut with a knife.
- Cadmium usually occurs
in deposits with zinc, such as sphalerite, and is recovered by a
roasting of
the mixed ore. It is a by product of zinc
refining.

- Cadmium is used in
electroplating, and in the production of bearing
alloys.
- The best known use of
Cadmium is in NiCad rechargeable batteries though their disposal poses an
environmental problem.
-
Artists have long used
the sulfide as a pigment – Cadmium Yellow.

- Cadmium is used as a
barrier to control atomic fusion.
- The sulfide and
selenide of cadmium have been used as phosphors in color television tubes.
- Most types of
food
(traditional and market meats, vegetables, breads, dairy, etc.) contain some
cadmium, but organs (livers and kidneys) have the highest levels.
- Smoke from cigarettes
contains cadmium and smokers have more cadmium in their
blood than
nonsmokers.
- Long term exposure to
high levels of cadmium can cause permanent kidney damage.
- Eating food or drinking
water with very high cadmium levels severely irritates the stomach, leading to
vomiting and diarrhea. The only people who have died from drinking cadmium are
people who used cadmium to commit suicide.



![]()

 Cadmium was
discovered by Stromeyer in 1817 as an impurity in a zinc carbonate ore. Stromeyer noted the yellow color of a specific batch of cadmia and found
cadmium as the contaminant.
Cadmium was
discovered by Stromeyer in 1817 as an impurity in a zinc carbonate ore. Stromeyer noted the yellow color of a specific batch of cadmia and found
cadmium as the contaminant.